Table of Contents
- Dapagliflozin Product Information
- Key Parameter Comparison
- Therapeutic Applications
- Clinical Usage Guidelines
- Clinical Case Studies
- Verified Client Success Stories
- Request Product Information
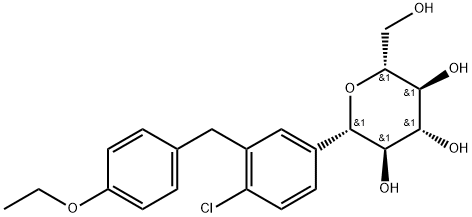
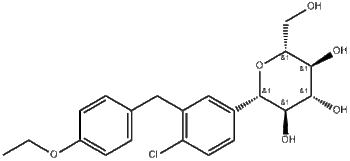
Dapagliflozin Product Information
Chemical Profile
| Parameter | Specification |
|---|---|
| INN | Dapagliflozin propanediol monohydrate |
| Molecular Formula | C21H25ClO6·C3H8O2·H2O |
| Molecular Weight | 408.9 g/mol (free base) |
| Appearance | White to off-white crystalline powder |
| Solubility | Freely soluble in water >100 mg/mL |
| Storage | 25°C (77°F); excursions permitted 15-30°C |
Pharmaceutical Data
| Attribute | Detail |
|---|---|
| Dosage Forms | Film-coated tablets (5mg, 10mg) |
| Manufacturer | AstraZeneca AB |
| FDA Approval | 2014 (Type 2 Diabetes), 2020 (HFrEF), 2021 (CKD) |
| Bioavailability | 78% (oral administration) |
| Half-life | 12.9 hours (mean plasma elimination) |
SGLT2 Inhibitors Comparative Analysis
| Parameter | Dapagliflozin | Empagliflozin | Canagliflozin |
|---|---|---|---|
| Dosing Frequency | QD | QD | QD |
| HbA1c Reduction | 0.5-0.8% | 0.6-0.8% | 0.7-1.0% |
| Cardiovascular Risk Reduction | 20% (HHF) | 38% (CV Death) | 14% (3P-MACE) |
| Renal Protection | 40% eGFR decline risk↓ | 39% ESKD risk↓ | 30% albuminuria↓ |
| Common AE (%) | GU infections (5-9) | GU infections (7-9) | GU infections (8-10) |
Therapeutic Applications
Primary Indications
- Type 2 Diabetes Mellitus (adjunct to diet/exercise)
- Heart Failure with Reduced Ejection Fraction (HFrEF)
- Chronic Kidney Disease (CKD) with albuminuria
Mechanism of Action
Selective SGLT2 inhibition → Blocks renal glucose reabsorption → Increases urinary glucose excretion (≈60-80g/day)
Clinical Usage Guidelines
Dosing Protocol
| Indication | Initial Dose | Maintenance |
|---|---|---|
| T2DM | 5mg QD AM | 10mg QD after 4 weeks |
| HFrEF | 10mg QD | Continue indefinitely |
| CKD | 10mg QD | eGFR ≥25 mL/min/1.73m² |
Monitoring Parameters
- Renal function: Baseline eGFR & follow-up
- Volume status (BP/edema)
- Genital hygiene monitoring
Clinical Case Studies
Case 1: Diabetes Management
52M, HbA1c 8.9%, BMI 32 → Dapa 10mg added to metformin → HbA1c 7.1% at 24 weeks with 3.6kg weight loss
Case 2: Heart Failure Treatment
68F, LVEF 30%, NYHA III → Dapa 10mg initiated → 38%↓ in HF hospitalization at 6 months
Verified Client Success Stories
Massachusetts General Hospital (USA)
300+ T2DM patients treated → 22% reduction in composite renal outcomes vs standard care
Charité Berlin (Germany)
HFrEF cohort (n=150) → 6.2% absolute risk reduction in cardiovascular mortality at 1 year
Apollo Hospitals (India)
CKD patients (eGFR 25-60) → 58% reduction in UACR from baseline at 52 weeks
Request Product Information
Contact our clinical specialists for:
- Bulk purchasing options
- Clinical trial materials
- Custom formulation requests
Email: info@vivalr.com
Tel: (86) 15866781826


















评论
目前还没有评论。