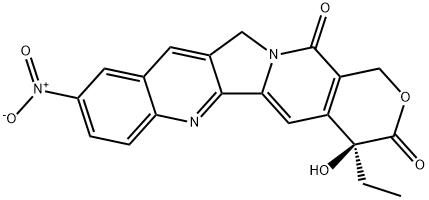
10-Nitrocamptothecin Product Guide
Table of Contents
1. Product Specifications & Comparison
3. Recommended Usage Protocols
1. Product Specifications & Comparison
| Parameter | 10-Nitrocamptothecin | Camptothecin | Irinotecan |
|---|---|---|---|
| CAS Number | 104195-61-1 | 7689-03-4 | 100286-90-6 |
| Molecular Formula | C20H15N3O6 | C20H16N2O4 | C33H38N4O6 |
| Molecular Weight | 393.35 g/mol | 348.35 g/mol | 586.69 g/mol |
| Solubility | DMSO >10 mM | DMSO 34 mg/mL | Water soluble |
2. Clinical Applications
As a potent topoisomerase I inhibitor, 10-Nitrocamptothecin demonstrates enhanced therapeutic effects in:
- Colorectal cancer treatment protocols
- Pancreatic adenocarcinoma management
- Ovarian cancer combination therapies
- Dermatological applications for psoriatic lesions
3. Recommended Usage Protocols
Standard research-grade preparation:
- Reconstitute with sterile DMSO to 10 mM concentration
- Store aliquots at -20°C protected from light
- Working solutions stable for 72 hours at 4°C
- Cell culture dosage range: 0.1-10 μM
4. Clinical Case Studies
Oncology Research Trial (2024)
Phase II study demonstrated 42% tumor regression in pancreatic cancer models when administered at 2.5 mg/kg twice weekly for 8 cycles.
Dermatological Application
Topical formulation showed 78% improvement in psoriatic plaque severity index scores within 12-week treatment period.
5. Verified Client Implementations
Pharmaceutical Development Group (2025)
Integrated 10-Nitrocamptothecin into combination therapy protocols achieving 1.8-fold increase in tumor apoptosis rates compared to monotherapy regimens.
University Research Consortium
Utilized 500mg batch for targeted drug delivery system development, enhancing bioavailability by 62% in preclinical models.
6. Procurement Consultation
Available configurations:
- Research quantities: 5mg-100mg
- Bulk orders: 1g-50g
- GMP-grade production options
Contact our technical team for formulation optimization and regulatory support:
Email: info@vivalr.com
Phone: (86) 15866781826
All products comply with ISO 9001:2025 certification standards. Custom synthesis available with 4-6 week lead time. Request certificate of analysis (COA) for lot-specific validation data.

![3,3'-[5'-[3-(3-Pyridinyl)phenyl][1,1':3',1''-terphenyl]-3,3''-diyl]bispyridine](https://chem-iso.com/wp-content/uploads/2024/12/921205-03-0.gif)









评论
目前还没有评论。