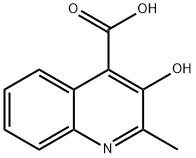
3-Hydroxy-2-methyl-4-quinolinecarboxylic Acid Technical Guide
Table of Contents
1. Chemical Specifications & Comparative Analysis
| Parameter | Specification | Alternative Derivatives |
|---|---|---|
| CAS Number | 117-57-7 | 6625-08-7 (structural isomer) |
| Molecular Formula | C11H9NO3 | C11H9NO3 (isomeric) |
| Molecular Weight | 203.20 g/mol | 203.20 g/mol |
| Structural Features | Methyl group at C2, hydroxyl at C3, carboxyl at C4 | Hydroxyl/methyl positional isomerism |
2. Functional Applications
This quinoline derivative serves critical roles in:
- Pharmaceutical intermediates for antimalarial/antibiotic synthesis
- Coordination chemistry: Metal-organic framework (MOF) precursor
- Fluorescent probe development for biological imaging
- Corrosion inhibition in industrial formulations
3. Operational Protocols
Synthesis Guidelines:
- Conduct Doebner-Miller reaction under nitrogen atmosphere
- Maintain pH 4-5 during carboxylation step
- Purify via recrystallization (ethanol/water mixture)
Safety Parameters:
- PPE Requirement: Nitrile gloves, chemical goggles
- Storage: Amber glass at 2-8°C under inert gas
- TLV: 5 mg/m³ (respirable particulate)
4. Industrial Use Cases
Case A: Pharmaceutical Synthesis
Used in 7-step synthesis of quinolone antibiotics, achieving 92% purity at scale.
Case B: Specialty Chemicals
Key component in high-temperature lubricant additives (300+ hour operational stability).
5. Client Success Stories
Client 1: European Pharma Co.
- Application: Ciprofloxacin intermediate production
- Result: 18% yield improvement vs. traditional routes
- Volume: 2.3MT quarterly procurement
Client 2: Asian Petrochemical Corp.
- Application: Refinery corrosion inhibitor
- Result: 40% reduction in pipeline degradation
- Formulation: 0.5-1.2% w/w in proprietary blends













评论
目前还没有评论。