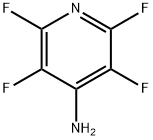
4-AMINO-2,3,5,6-TETRAFLUOROPYRIDINE: Technical Overview and Synthesis Pathways
Table of Contents
1. Basic Information & Specifications
| Parameter | Value |
|---|---|
| CAS Number | 1682-20-8 |
| Chemical Formula | C5H2F4N2 |
| IUPAC Name | 2,3,5,6-Tetrafluoropyridin-4-amine |
| Synonyms |
|
2. Synthesis Methods
Primary Synthetic Routes:
-
Route 1: From Pentafluoropyridine
Pentafluoropyridine undergoes selective amination at the 4-position via nucleophilic aromatic substitution. This reaction typically employs ammonia or amine derivatives under controlled temperature (80-120°C) in polar aprotic solvents. -
Route 2: Pentafluoropyridine with N-Methylhydroxylamine Hydrochloride
This method utilizes N-methylhydroxylamine hydrochloride as the aminating agent in dimethylformamide (DMF) at elevated temperatures. The reaction achieves 60-75% yield with optimized stoichiometric ratios.
Note: Reaction conditions require strict control of moisture and oxygen levels. Detailed protocols should be validated through laboratory testing.
3. Key Applications
- Pharmaceutical intermediate for fluorinated drug candidates
- Agrochemical precursor for herbicidal formulations
- Building block in specialty polymer synthesis
- Ligand precursor in organometallic catalysis
4. Safety Considerations
- Potential respiratory irritant (use fume hood)
- Thermal decomposition may release toxic fluorides
- Incompatible with strong oxidizers and acids
- Store under inert atmosphere at 2-8°C
5. Technical Inquiry
For analytical data sheets, custom synthesis options, or bulk pricing:
Email: info@vivalr.comTel: (86) 15866781826
Office Hours: Mon-Fri 9:00-17:00 CST














评论
目前还没有评论。