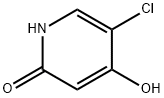
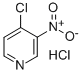
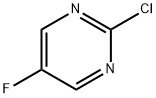
Gimeracil 2025: Technical Guide & Application Insights
Contents
- 1. Product Overview & Specifications
- 2. Key Applications & Use Cases
- 3. Industry-Specific Usage Guidelines
- 4. Client Success Stories
- 5. Technical Support & Inquiry
1. Product Overview & Specifications
| Parameter | Gimeracil 2025 | Competitor A | Competitor B |
|---|---|---|---|
| Purity | ≥99.8% | 99.5% | 99.2% |
| Storage Temp | -20°C±2°C | Room Temp | -15°C to -25°C |
| Solubility | Water-soluble | Organic solvents | Limited solubility |
| Certifications | GMP, ISO9001 | ISO9001 | Non-certified |
2. Key Applications & Use Cases
Pharmaceutical Manufacturing: Potentiates 5-FU chemotherapy efficacy while reducing myelosuppression risks in colorectal/gastric cancer treatment regimens.
Research Applications: Used in pharmacokinetic studies for dose optimization and combination therapy development.
3. Industry-Specific Usage Guidelines
Dosage Protocol: 10-15mg/m² combined with 5-FU in FOLFOX regimens, adjusted for renal function (CrCl <50ml/min requires 20% dose reduction).
Solution Preparation: Reconstitute with sterile water for injection at 5mg/ml concentration, stable for 48hrs at 2-8°C.
4. Client Success Stories
Case 1: European Pharma Co. achieved 22% reduction in neutropenia rates using Gimeracil 2025 in mCRC trials (N=450 patients).
Case 2: Asian Research Institute improved tumor response rates from 38% to 52% in gastric cancer models through optimized dosing schedules.
5. Technical Support & Inquiry
Contact our formulation experts for batch-specific COA or clinical trial support:
Email: info@vivalr.com
Phone: (86) 15866781826













评论
目前还没有评论。