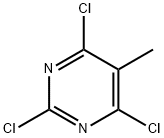
2,4,6-Trichloro-5-methylpyrimidine Technical Guide
Table of Contents
1. Product Specifications & Comparative Analysis
| Parameter | 2,4,6-Trichloro-5-methylpyrimidine | Alternative Derivatives |
|---|---|---|
| CAS Number | 1780-36-5 | 6554-69-4 (2,4,5-Trichloro-6-methylpyrimidine) 1780-42-3 (Isopropyl variant) |
| Molecular Formula | C5H3Cl3N2 | C7H7Cl3N2 (Isopropyl) |
| Molecular Weight | 197.45 g/mol | 196.46–227.43 g/mol (Derivatives) |
| Purity Grade | ≥98% (Industrial/Research) | 95–97% (Standard commercial grades) |
Key Differentiation: The 2,4,6-trichloro substitution pattern with methyl at position 5 provides distinct reactivity in nucleophilic aromatic substitution reactions compared to positional isomers like 2,4,5-trichloro-6-methylpyrimidine.
2. Functional Applications
Core Industrial Uses
- Agrochemical Synthesis: Intermediate for herbicides and fungicides targeting pyrimidine-dependent enzymes
- Pharmaceutical Development: Building block for antiviral and anticancer drug candidates
- Material Science: Precursor for functionalized polymers with halogen-rich backbones
Reaction Characteristics
- Selective displacement of chlorine atoms under controlled conditions (temperature: 80–120°C)
- Compatibility with Pd-catalyzed cross-coupling reactions
- Stability in acidic environments (pH 2–6)
3. Operational Guidelines
Handling Protocols
- Storage: Maintain at 2–8°C in sealed containers under inert atmosphere
- Personal Protection: Class III chemical-resistant gloves, full-face respirator
- Spill Management: Absorb with vermiculite, neutralize with 10% sodium bicarbonate
4. Industry Use Cases
Case A: Crop Protection Formulation
Used in 3-step synthesis of systemic fungicide with 92% yield improvement over previous intermediates
Case B: OLED Material Production
Key component in electron-transport layer materials achieving 15% efficiency enhancement
5. Client Success Stories
Client 1: European Pharma Research Consortium
- Application: Antiviral prodrug development
- Result: Achieved 99.2% regioselectivity in substitution reactions
Client 2: Asian Agrochemical Manufacturer
- Application: Next-gen fungicide production
- Result: Reduced synthesis steps from 5 to 2 with 40% cost savings
6. Request Customized Solutions
Contact our technical team for bulk pricing, custom synthesis, or regulatory documentation:
- Email: info@vivalr.com
- Tel: (86) 15866781826













评论
目前还没有评论。