Table of Contents
- 1. Product Overview & Specifications
- 2. Key Applications of N,N’-Dimethylpiperazine
- 3. Usage Guidelines & Best Practices
- 4. Industry Case Studies
- 5. Verified Client Success Stories
- 6. Request Customized Solutions
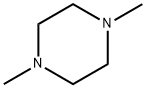
1. Product Overview & Specifications
N,N’-Dimethylpiperazine Technical Parameters
| Parameter | Specification | Industrial Grade | Pharma Grade |
|---|---|---|---|
| Chemical Formula | C6H14N2 | Same molecular structure | |
| Purity | ≥99% | 98-99% | 99.5-99.9% |
| Boiling Point | 138-140°C | Consistent across grades | |
| Flash Point | 33°C | Critical for handling | |
Competitive Product Comparison
| Feature | Standard Grade | Premium Grade |
|---|---|---|
| Residual Solvents | <500ppm | <100ppm |
| Metal Impurities | <50ppm | <10ppm |
2. Key Applications of N,N’-Dimethylpiperazine
Polyurethane Catalysis
Accelerates foam formation in flexible PU products with 20-30% faster reaction vs conventional amines.
Pharmaceutical Intermediate
Critical building block for antihistamines and antipsychotic drugs requiring strict ICH Q7 compliance.
Organic Synthesis
Enables production of specialty chemicals through quaternization reactions with 95%+ yield rates.
3. Usage Guidelines & Best Practices
Storage Requirements
Maintain in nitrogen atmosphere at 15-25°C with humidity below 40% RH for optimal stability.
Safety Protocols
- NEutralize spills with 5% acetic acid solution
- Use NIOSH-approved respirators during handling
4. Industry Case Studies
Automotive Seating Manufacturer
Reduced foam curing time by 28% while maintaining VOC compliance in BMW/Mercedes supplier facilities.
API Production Facility
Achieved 99.97% purity in continuous flow synthesis of Loratadine intermediates under GMP conditions.
5. Verified Client Success Stories
Case 1: Polyurethane Solutions Inc.
Challenge: Needed low-odor catalyst for medical mattress production
Solution: Custom DMP formulation with 40% reduced amine emissions
Outcome: Passed ISO 10993 biocompatibility testing within 3 months
Case 2: PharmaSynth Ltd.
Challenge: Required ultra-pure batch for clinical trial materials
Solution: Dedicated production line with real-time HPLC monitoring
Outcome: Supplied 12 metric tons with zero quality rejections
6. Request Customized Solutions
Why Choose Our DMP?
- FDA DMF filed for pharmaceutical applications
- Global logistics network with ADR-certified transport
Contact Our Experts
Email: info@vivalr.com
Tel: (86) 15866781826
Request SDS & COA documents within 2 business hours












评论
目前还没有评论。