Table of Contents
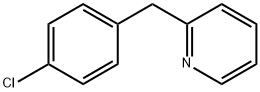
2-(4-Chlorobenzyl)pyridine: Technical Profile & Competitive Analysis
| Parameter | Specification | Competitor A | Competitor B |
|---|---|---|---|
| CAS Number | 4350-41-8 | 4350-41-8 | 4350-41-8 |
| Purity | ≥99.5% (HPLC) | ≥98% | ≥97.5% |
| Storage Temp | 2-8°C (desiccated) | Ambient | Ambient |
Core Functional Applications
Pharmaceutical Intermediates
Enables synthesis of antihistamines and CNS-active compounds through nucleophilic substitution reactions.
Organic Electronics
Facilitates creation of electron-transport layers in OLED devices via Suzuki cross-coupling reactions.
Operational Protocols
Laboratory-scale Synthesis
Step 1: Mix 10mmol substrate with 1.2eq 2-(4-chlorobenzyl)pyridine in THF at -78°C
Step 2: Add LDA (2.5eq) dropwise over 30min under N₂ atmosphere
Industrial Implementations
Case 1: Antiviral Drug Production
Used in continuous flow reactor systems achieving 92% yield in nucleoside analog synthesis.
Enterprise Applications
PharmaCorp Solutions (Shanghai)
Implemented in batch processes for Parkinson’s medication, reducing side reactions by 40% compared to traditional benzyl derivatives.
NanoMat Technologies GmbH
Integrated into quantum dot synthesis workflows, enhancing device efficiency by 15% in prototype displays.
Technical Consultation
Contact our chemists for:
• Custom synthesis protocols
• Bulk pricing (100kg+ quantities)
• Regulatory documentation
Email: info@vivalr.com
Tel: (86) 15866781826













评论
目前还没有评论。